What if the device tracking your heart rate, hydration, or lactate levels during a run could power itself entirely from your sweat — no battery, no charging cable, no interruption? That’s not a distant concept anymore. Researchers in Japan say they’ve found a cleaner, simpler way to manufacture exactly that kind of self-powered wearable sensor.
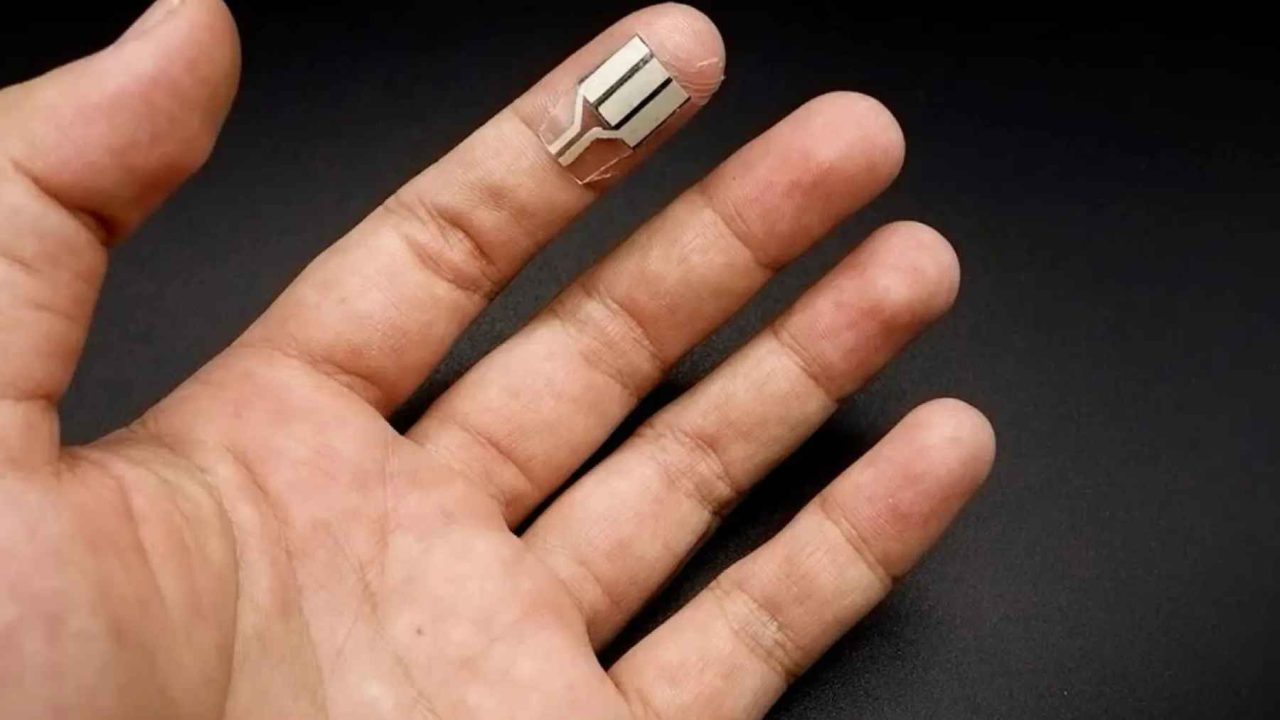
The team has developed a water-based “enzyme ink” that can be printed onto paper in a single step, creating a sensor capable of generating electricity from the lactate in human sweat. Their results, published online on February 6, 2026, in ACS Applied Engineering Materials, point to a peak power density of about 165 microwatts per square centimeter — roughly 1,065 microwatts per square inch — at 0.63 volts.
For anyone who has ever watched a fitness tracker die mid-workout, the implications are hard to ignore.
How a Patch on Your Skin Could Replace a Battery
The devices at the center of this research are called enzymatic biofuel cells, or EBFCs. The concept isn’t entirely new — scientists have been exploring biofuel cells for years — but manufacturing them cleanly and cheaply enough for real-world wearable use has remained a persistent challenge.
What the Japanese team has done differently is develop a water-based enzyme ink that simplifies the production process down to a single printing step onto paper. That’s a meaningful shift. Most earlier approaches required more complex fabrication methods, which made scaling up difficult and raised questions about cost and practicality.
The basic operating principle is straightforward. Enzymes embedded in the printed material help pull electrons from lactate — a compound naturally present in sweat, especially during physical activity. That electron movement generates an electrical current. The sensor doesn’t just power itself; it also measures lactate concentration across a range of roughly 1 to 25 millimolar, which the researchers say corresponds to real exercise conditions.
That dual function — power generation and metabolic monitoring at the same time — is what makes this design particularly interesting for wearable health technology.
The Numbers Behind the Breakthrough
The performance figures reported by the team give a clearer sense of what this technology can actually deliver. Here’s a breakdown of the key technical details confirmed in the published research:
| Metric | Value |
|---|---|
| Peak power density | ~165 microwatts per cm² (~1,065 microwatts per in²) |
| Operating voltage | 0.63 volts |
| Lactate detection range | ~1 to 25 millimolar |
| Publication date | February 6, 2026 |
| Published in | ACS Applied Engineering Materials |
| Manufacturing method | Single-step water-based enzyme ink printing on paper |
The lactate range of 1 to 25 millimolar is worth paying attention to. Lactate levels in sweat rise as exercise intensity increases, making it a useful real-time marker of physical exertion and metabolic stress. A sensor that can track that range while simultaneously powering itself covers a meaningful slice of what athletes and health monitors actually need to measure.
- The ink is water-based, making it less reliant on harsh chemical processes during manufacturing
- Printing onto paper keeps the substrate flexible, lightweight, and low-cost
- The single-step process reduces production complexity compared to earlier EBFC fabrication methods
- The fuel source — lactate in sweat — is continuously produced by the body during exercise, meaning the sensor activates naturally when it’s needed most
Why This Matters for Wearable Health Technology
Wearable health devices have become genuinely useful tools for tracking fitness, managing chronic conditions, and catching early warning signs. But nearly all of them share the same fundamental weakness: they run on batteries that eventually die, need to be recharged, and add bulk and weight to the device.
Self-powered biosensors would sidestep that problem entirely. A patch that draws energy from the wearer’s own body fluids could, in theory, run continuously without any external power source. That’s especially relevant for applications where consistent, uninterrupted monitoring matters — think post-surgical recovery tracking, athletic performance monitoring, or long-term management of conditions like diabetes where metabolic markers need regular observation.
The paper-based, single-step manufacturing approach also hints at lower production costs, which matters if this technology is ever going to move beyond laboratory demonstrations and into affordable consumer or clinical products.
Researchers point out that enzymatic biofuel cells have long held promise for exactly this kind of application, but manufacturing complexity has been a barrier. A simpler, cleaner fabrication method — using materials that don’t require elaborate chemical handling — is one of the more practical steps toward closing that gap.
Where This Research Goes From Here
The study published in February 2026 represents a proof of concept — a demonstration that the enzyme ink approach works and that the performance numbers are competitive. What comes next is a longer road.
For a technology like this to appear in a commercial wearable, researchers and manufacturers would need to confirm that the sensors perform consistently across different users, different sweat compositions, and different environmental conditions. Durability over extended wear periods, skin compatibility, and the ability to integrate with data-reading electronics are all factors that lab results alone don’t fully answer.
What the research does establish is that the manufacturing method is viable, the power output is measurable and meaningful, and the sensing range covers real physiological conditions.
For a field that has been working toward self-powered wearables for years, that’s a concrete step forward — even if the finish line is still some distance away.
Frequently Asked Questions
What is an enzymatic biofuel cell (EBFC)?
An enzymatic biofuel cell generates electricity by using enzymes to drive chemical reactions in body fluids, such as sweat. In this research, the main fuel is lactate produced during exercise.
How much power does the sweat-powered sensor generate?
The researchers reported a peak power density of approximately 165 microwatts per square centimeter, or around 1,065 microwatts per square inch, at 0.63 volts.
What does this sensor actually measure?
The sensor measures lactate concentration in sweat across a range of roughly 1 to 25 millimolar, a range the researchers say corresponds to real exercise conditions.
What makes this manufacturing method different from earlier approaches?
The team uses a water-based enzyme ink printed onto paper in a single step, which simplifies the production process compared to more complex fabrication methods used in earlier EBFC research.
When was this research published?
The findings were published online on February 6, 2026, in the journal ACS Applied Engineering Materials.
Is this technology available in consumer products yet?
This has not been confirmed. The published research represents a proof of concept, and the path to commercial wearable products would require additional testing, durability studies, and development work beyond what the current study addresses.





Leave a Reply