Aluminum is the most abundant metal in Earth’s crust, used in everything from soda cans to aircraft fuselage panels. But for all its familiarity, scientists at King’s College London have just revealed a version of it that behaves in a way nobody had seen before — and the implications could stretch far beyond the laboratory.
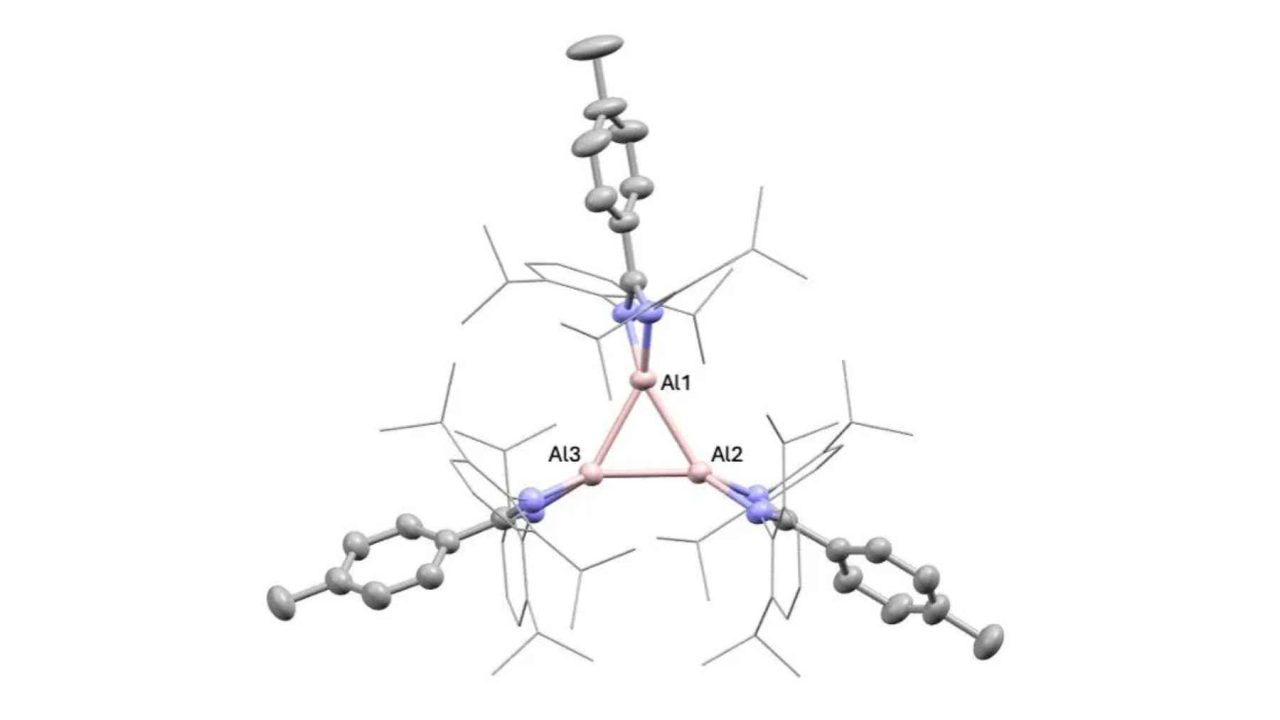
The researchers have created a three-atom ring of aluminum, a structure called a cyclotrialumane, that displays what the team describes as “unprecedented reactivity.” That phrase alone is enough to make chemists sit up. In a field where incremental progress is the norm, a material doing something genuinely new — especially one made from one of the cheapest and most widely available metals on Earth — is worth paying close attention to.
The research was published in Nature Communications, one of the most respected scientific journals in the world, and it arrives at a moment when industries across the globe are scrambling to reduce their dependence on rare, expensive, and geopolitically sensitive critical metals.
What Scientists Actually Built — and Why It’s Unusual
To understand why this matters, it helps to know what aluminum usually does. In everyday life, aluminum is stable, lightweight, and predictable. It doesn’t tend to behave like a reactive chemical agent. That stability is exactly what makes it useful in packaging and construction — and exactly what makes this new discovery so surprising.
The compound the King’s College London team created is described as a neutral aluminum(I) trimer — essentially, a tiny triangular cluster of three aluminum atoms bonded together in a ring. The formal name is cyclotrialumane. What sets it apart isn’t just the structure, but the behavior: this triangular arrangement stays intact when dissolved in solution, which is a critical requirement for any compound you want to use as a practical chemical tool.
According to the Nature Communications paper, neutral trimeric aluminum(I) structures had been “notably absent” from previous research. The team reports two examples of this compound, both of which retained their trimeric form in solution — a key detail that separates interesting lab curiosities from genuinely useful chemistry.
The aluminum in this compound is in what chemists call the +1 oxidation state, written as aluminum(I). This is far less common than the +3 state you find in standard aluminum compounds. That lower oxidation state is part of what gives the material its unusual reactivity, making it behave more like a sophisticated chemical tool than a passive structural material.
The Connection to Critical Metals — and Why It Matters Now
Here is where the story connects to something much bigger than chemistry.
Right now, industries building the clean energy future — electric vehicles, fuel cells, industrial chemical processes — depend heavily on a group of metals known as critical metals. These include platinum, palladium, and similar elements. They are exceptional catalysts, meaning they speed up chemical reactions without being consumed in the process. But they are also rare, expensive, difficult to source, and concentrated in a small number of countries, creating serious supply chain vulnerabilities.
The new aluminum compound hints at the possibility that, someday, a far more abundant and affordable material could take on some of those roles. The researchers describe the cyclotrialumane’s reactivity as comparable in character — at least in exploratory terms — to what we typically expect from far rarer metals.
This is still early-stage science. Nobody is claiming aluminum will replace platinum in a fuel cell next year. But the direction of the research points squarely at one of manufacturing’s most pressing problems: how to build a cleaner, more sustainable industrial base without depending on materials that are scarce, costly, and vulnerable to supply disruption.
Key Facts About the Discovery at a Glance
| Detail | What the Research Shows |
|---|---|
| Institution | King’s College London |
| Published in | Nature Communications |
| Compound type | Neutral aluminum(I) trimer (cyclotrialumane) |
| Structure | Three-atom aluminum ring |
| Key property | Trimeric form retained in solution |
| Reactivity described as | “Unprecedented” by the research team |
| Number of examples reported | Two |
| Prior status of this structure | “Notably absent” from earlier scientific work |
- The compound is described as neutral, meaning it carries no overall electrical charge — an unusual trait for highly reactive aluminum species.
- The aluminum(I) oxidation state is far rarer in stable compounds than the aluminum(III) state found in common materials.
- Stability in solution is a practical requirement for catalytic applications, making this a meaningful milestone beyond a purely structural curiosity.
- The research is positioned within the broader scientific effort to develop catalysts that do not rely on critical or precious metals.
Who Stands to Benefit If This Research Scales
If the science behind cyclotrialumane can be developed further, the potential beneficiaries are widespread. Chemical manufacturers who currently pay premium prices for platinum-group metal catalysts would have reason to watch this closely. So would the clean energy sector, where catalytic processes are central to hydrogen production, fuel cells, and emissions reduction technologies.
Aluminum is not just abundant — it is already one of the most recycled materials on the planet, with established global supply chains and relatively low extraction costs compared to rare earth or precious metals. A catalytic aluminum compound that could substitute for rarer materials, even in a narrow range of applications, would represent a meaningful shift in industrial economics.
For consumers, the downstream effect could eventually mean lower costs in products and technologies that currently depend on expensive rare metal components. That is a long chain of development from a laboratory compound to a manufactured product, but every such shift starts with exactly this kind of foundational chemistry research.
What Comes Next for This Research
The King’s College London team’s paper represents an early but significant step. Reporting two confirmed examples of the cyclotrialumane structure, and demonstrating that the trimeric form holds in solution, gives other researchers a foundation to build on.
The next stages would typically involve testing the compound’s reactivity across a wider range of chemical reactions, investigating its stability under different conditions, and exploring whether it can be synthesized efficiently enough to be practical. None of that work has been reported yet, and the researchers themselves characterize this as exploratory science.
What is clear is that the field of low-oxidation-state aluminum chemistry has opened a new door. Whether what lies behind it transforms manufacturing or remains a fascinating footnote will depend on years of follow-on research — but the starting point is genuinely novel.
Frequently Asked Questions
What is a cyclotrialumane?
It is a neutral aluminum(I) trimer — a three-atom ring of aluminum that displays unusual and highly reactive chemical behavior, as reported by researchers at King’s College London in Nature Communications.
Why had this type of aluminum structure not been found before?
According to the research paper, neutral trimeric aluminum(I) structures had been “notably absent” from earlier scientific work, making these two reported examples the first of their kind.
Could this aluminum compound replace platinum or palladium in industrial use?
The research is exploratory and no direct replacement has been demonstrated. Scientists note the compound shows “unprecedented reactivity,” which raises long-term possibilities, but practical applications have not yet been confirmed.
Why does it matter that the compound stays intact in solution?
Stability in solution is a practical requirement for catalytic chemistry. A compound that breaks apart when dissolved cannot be reliably used as a chemical tool, so this property is a meaningful step toward potential real-world application.
Where was this research conducted and published?
The research was conducted at King’s College London and published in Nature Communications.
Is aluminum a realistic alternative to critical metals for industrial catalysis?
This research suggests it is worth investigating seriously, but the science is at an early stage. Aluminum’s abundance and established supply chains make it an attractive candidate if the chemistry can be developed further.




Leave a Reply